During the 2019 AMPLEXOR ‘Be The Expert’ meeting, we had the pleasure to present our views on the potential of structured content authoring for the Life Sciences industry.
We did so by comparing the adoption of ‘structured content’ in Life Sciences with the state of adoption in other industries. We notably compared Life Sciences with the following ‘leading’ industries: Media and Publishing, Manufacturing, Aviation, Hardware, and Software.
Structured content has had attention in Life Sciences for quite a while. Understandable, because the submission processes for medicine tick many boxes for structured content.
- Submissions have a strictly defined document structure;
- Within dossiers there is a high level of content repetition and reuse;
- Across markets, submissions are largely but not completely identical, making a case for automated document generation;
- Regulators are moving in the direction of digital, data-centric submissions;
- Documents are populated with data from other systems within the company; preferably done by automated content/data integration.
Despite all this, to date, structured content in Life Science companies has only reached limited implementation. When discussing similarities and differences between the various industries, a few observations came up:
- Life Science historically has had substantial budgets available. The cost reductions that structured content brings, have not been enough of an argument to outweigh the effort of change-management;
- Regulators are moving slower than expected in their evolution from document-driven to data-driven submissions. Formatted PDF, accompanied with metadata, is still the digital format that forms the core of most submissions.
- Typically, structured content projects (most of them in Proof of Concept stage) have been strongly IT-driven and received less support from business-side of companies.
Structured content — climbing Gartner’s slope towards productivity
As Gartner puts it in its 2018 Hype Cycle Report, Structured Content and Component-Based authoring are beginning to climb the slope toward productivity. It means that companies are now adding structured content capabilities to their IT-landscape — after expectations have first been inflated, then tempered and now are gradually growing.
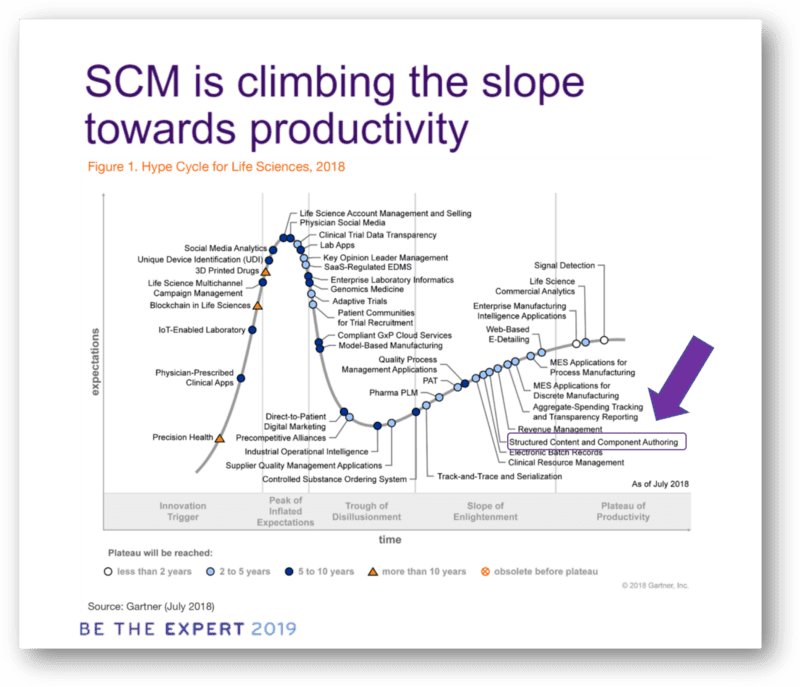
Vendors such as AMPLEXOR are integrating structured content capabilities in their end-to-end solutions, expecting that in the mid-term (2–5 years from now), the use of content-components from which documents are generated will indeed replace the document-centric practice of today.
One of the questions remaining is what the drivers behind wide adoption (plateau of productivity) will be. As concluded during the conference, the following developments will continue to push towards structured content authoring.
- The question is not if regulators will take further steps towards data-driven content submissions, but rather when and how exactly. A policy-event would likely accelerate structured content adoption.
- The rise of tailored medicine, making the number of variants of products that will be registered greatly rising, which increases the need for efficient submission.
- Tangible proof of how structured content can accelerate filing will move interest from cost and risk management towards reaching extra revenue.
Last but not least, technology has advanced and arrived at a level that makes structured content authoring feasible for non-XML experts. Most of the PoCs that pharma companies have done (5–10 years ago) have been conducted using authoring solutions that typically led to resistance from business stakeholders because of their complexity.
User-friendly solutions like Fonto, mostly embedded in an industry-specific workflow solution, have shown to be capable of onboarding authors in industries such as Media, Publishing, Manufacturing, Aviation, Regulatory and others. In Life Sciences, we feel that structured content will indeed enter the “Plateau of Productivity’ in the years to come.
CEO at Fonto. Enthusiastic sailor. Tweets in English and Dutch.
